Stone, minerals and semiprecious of the world stone
Carbonate: Dolomite -->rus
 Diagnostic cart.
Diagnostic cart.
On a photo: crystals of dolomite and Pyrite. Down: will break a secret from the mine of Traversella (Turin). Ball from the polycoloured (multicoloured) dolomite
Ca Mg (СO3)2
Crystal structure trigonal
Hardness on the Mohs scale 3,5-4
Specific unit weight mass 2,85-2,95
Cleavage divided easily
Fracture, break padman
Colors colourless, multicolors
Colors in powder triturate white
Glance (glitter, glare) from glass to mother-of-pearl
Mineral and consisting of him rock name a dolomite. Calcspar and magnesium. Glance (glitter, glare) glassy. Transparent. Colors: white, rather yellow, brownish; now and then is colourless and semilucent. A line is white. Fracture, break padman. Fragile. Cleavage perfect absolute. At contiguity with "boils" up warm dilute muriatic acid.
Porodoobrazuyuschiy mineral of carbonate siltages. Appears also in hydrothermal vein, lode, mines and cracks. Often be found in accretion with Calcite. Crystals (ditrigonalCrystal structure) saddle-like is bent usually. Rhombohedrons prevail among them (the Cleavage pricking out have a that form). Grainy aggregates, continuous, coarse-crystalline, macrocrystalline blocks, are not uncommon with clear expressed cleavage, dense and porous cellular the masses. Widespread everywhere.
Ankerite, or brown spar. Zhelezosoderzhaschiy dolomite. In large the masses be found rarely, usually appears in hydrothermal terms.
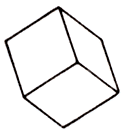
 Presented rhombohedrons, well-educated planes of verges, being rhombuses. The aggregates of crystals are typical also with the distorted surfaces of verges, givings them the look of "cavalry saddle".
Presented rhombohedrons, well-educated planes of verges, being rhombuses. The aggregates of crystals are typical also with the distorted surfaces of verges, givings them the look of "cavalry saddle".
Crystals, possessing good cleavage on a rhombohedron, can be colourless and transparent, sometimes light-grey, rather yellow, rose and then opaque. To brilliance of colourless Crystals-glassy, but mother-of-pear nacreous is more typical, especially at the saddle-shaped aggregates. A dolomite can be continuous the masses dense or saccharoidal additions, sometimes cavernous.
Chemical composition (chemistry, compound). Double salt of CaCO3 - MgSO3; kemidol (CaO) 30,4%, oxide of magnesium (MgO) 21,7%, dioxide of carbon (CO2) 47,9%; isomorphous admixtures: iron, manganese (to a few percents). Form of crystals. Rhombohedral; the verges of crystals are often distorted. Crystalline structure. Characterized that the ions of calcium (Ca) and magnesium (Mg) alternate along a triple ax. Class of symmetry. Rhombohedral - 3. Cleavage. Perfect on a rhombohedron (1011). Aggregates. Usually grainy, often porous, rarer kidney-shaped reniform, cellular, spherical.
 Diagnostic indication.
Diagnostic indication.
A dolomite scratches a knife and not too heavy. From Calcite differs that does not boil up at co-operating with dilute acids in-cold. Not melt fuse, but cracking. A behavior is in acids. In cold HCl dissolves slowly, and in warmed-up - quick (with the strong boiling up); Calcite strongly boils up already in cold HCl.
Origin provenance genesis.
Dolomite - widespread mineral, main component part of carbonate siltages, dolomites and dolomitized limestones. They appear mainly as a result of action of salt water, enriched magnesium, on the marine lime sinking. Thus there is a gradual substitution of part of calcium by magnesium. A dolomite is also in some low temperature hydrothermal vein, lode, mines among different metamorphic rocks.
Deposit minefield mine field occurrence subsoil.
In Italy widespread. In an area P'emont of crystal found out a dolomite in cracks among the sugar-like dolomites of Krevola-d'ossola. Magnificent crystals are known from the mineries of Brosso and Traversella near Turin. In an area Lombardy will break a secret as the saddle-shaped crystals discovered in quartz porphyries in Guasso-al'-monte (province Varese). A-plenty be found in area of dolomite structure, and also often be found among granular limestone, marble Karrary. The excellent standards of crystals of dolomites are known in a region Navarra (Spain).
Use, practical application, deployment.
A clean pure dolomite is excellent raw material for extraction of metallic magnesium, applied mainly at the production of easy alloys. From dolomites get salts of magnesium, in-use in medicine. In industry of build materials a dolomite is applied for making of the special brands of cement.

Magnesium is a component of enzymes, in the organism of people contained in bones, teeth, is the regulator of work of the nervous system. Magnesium is a macronutrient, from insufficiency of which to 90% people suffer. There is about 140 g of magnesium (0,2% from mass of body) in the organism of people, thus 2/3 is on bone tissues. A main "depot" of magnesium is in bones and muscles as phosphates and bicarbonate. Magnesium enters organism with food (in particular, with culinary salt - tastes bitter, "Artemsol'") and water.
Impoverishment of blood magnesium is marked at a rachitis. Signs of insufficiency of magnesium: worsening of transmission of nervous and muscular impulses, defiant crabbiness and nervousness, hypererethism, spasms and cramps, chronotaraxis and space, insomnia, migraine, chronic fatigue, violations of digestion, cardiopalmus, attacks, bursts of anger or irritation. The first sign of insufficiency of magnesium are twitches and cramps, especially gastrocnemius muscles. Psychiatric mineral.
Insufficiency of magnesium is underlaid cardiovascular diseases. The risk of origin of atherosclerosis of arteries of heart, cardiac attacks and changes of rhythm of heart rises on a background the permanent deficit of magnesium. It can be reason of lethal cardiac arrhythmia, hypertension, sudden stop of heart, asthma (especially on soil of breathing in a cinnabar), chronic pain syndrome (posttraumatic syndrome), depression, insomnia, syndrome of angry colon and pulmonary diseases. Arthritis and osteoporosis develops. Most magnesium is contained in wheat brans and germ of cereals (520 and 270 mgs on 100 g).
Magnesium is needed: at hypertensive illness, symptomatic hypertension, atherosclerosis, pathology of hepar and bilious ways, depression, dizzinesses, muscularweakness and convulsive reductions, at psoriasis, scleriasis, red lupus, premenstrual syndrome, for maintenance in the organism of normal pH balance, warning of calcinosis of soft tissues. It provides protecting of endothelia of arteries from stress, caused the sudden overfalls of arteriotony, needed for formation of bone tissues, carbohydrate and mineral exchange, for dissolution of kidney stone, formed from oxalate and phosphates. Magnesium is strengthened by efficiency of action of vitamin of V6 (pyridoxin), important for the prophylaxis of development of atherosclerosis.
The increase of level of magnesium (hypermagnesemia) in blood can take a place at the reception of antacid or laxatives, containing magnesium, at patients with chronic kidney insufficiency. At parenterally introduction of sulfate of magnesium there can be symptoms of intoxication as a general oppressing, languor and somnolence (a brain is involved), cardiac insufficiency. Application of solution of sulfate magnesia at pregnancy and births in 4 times increases the risk of development of DCP at new-born (weak births of mother). Anesthesia (to the coma) comes at the concentrations of magnesium in blood of 15-18 мг%.
Doctors are afraid of magnesium even, it is dangerous "comatose" ("sleepy") preparation, oppressive a cerebrum - practically 100% permanent establishment (at treatment medical magnesia). Mineral the ions of which "work" on the joint of neurons (axons). At sharp kidney insufficiency with oliguria, especially in combination with metabolic acidosis, hypermagnesemia is possible. Infuziya of calcium (antagonist) can counteract toxicness of magnesium.
DOPOG #4.1 
Flammable hard solid substance matters, self-reactive matters and solid desensitized explosives
Risk of fire. Flammable or combustible matters can catch a fire from sparks or flame. Can contain self-reactive matters, apt at exothermic decomposition in the case of heating, contact with other matters (such as: acids, connections of heavy metals or amines), to the friction or blow.
It can result in the selection of harmful or flammable gases or pair or spontaneous combustion. Capacities can burst at heating (dangerous, perilous - does not burn practically).
Risk of explosion of the desensitized explosives after the loss of desensitizer
Seven vertical red striaes on a white background, isometric, number of DOPOG, black flame
DOPOG #4.2 
Matters substances, apt at spontaneous combustion
Risk of fire as a result of spontaneous combustion in case if packing is damaged or the source of maintenance happened.
Can stormily react with water
White overhead half of rhombus, red - lower, isometric, number of DOPOG, black flame
DOPOG #4.3 

Matters substances, which select flammable gases at a contact with water
Risk of fire and explosion at a contact with water.
Gruz, which had scattered, it is necessary to cover and hold dry
Sine-blue rhombus, number of DOPOG, black or white flame
DOPOG #5.1 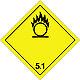
Matters substances, which oxidize
Risk of stormy reaction, self-ignition or explosion at a contact with combustible or flammable matters
To shut out formation of mixture of load with flammable or combustible matters (for example by sawdusts)
Yellow rhombus, number of DOPOG, black flame above by a circle
DOPOG #6.1 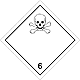
Toxic non biological matters substances (toxin)
Risk of poisoning at inhalation, contact with a derma or swallowing. Make a danger for a water environment or sewage system
To use a mask for emergency abandonment of transport vehicle
White rhombus, number of DOPOG, black skull and crossed cross-bones
