Stone, minerals and semiprecious of the world stone
Sulphate: Celestine (celestite) -->rus
 Diagnostic cart.
Diagnostic cart.
On a photo: plate tablet lamellar crystals of celestine. Down: ordinary type of mineral as the strong extended crystals.
Sr SO4 (sulfate of strontium)
Crystal structure monoclinic
Hardness on the Mohs scale 2
Specific unit weight mass 2,3-2,4
Cleavage perfect absolute
Fracture, break wrong
Colors colourless, various
Colors in powder triturate white
Glance (glitter, glare) from glass to mother-of-pearl
Celestine (celestite) is a sulfate of strontium. Hardness on the Mohs scale 3-3,5. Closeness 3,9-4,0. Glance (glitter, glare) mother-of-pear nacreous to glass. Transparent to translucent. It is colourless or white, but differences, painted in a light greyish-blue sky color, are not uncommon. A line is white. Fracture, break uneven. Fragile. Cleavage perfect absolute. Paints flame in a red color. Appears in cracks and drusy cavities. Crystals (rhombic Crystal structure) are rich in verges. Are columnar, basaltiform, plate tablet lamellar, there are grainy aggregates. Places of distribution: North Rhine-Vestfaliya (Germany), Yen (Germany), Zalcburg (Austria), Sicily (Italy), England, Mexico, CIS.
Presented extended-prismatic or (much rarer) by plate tablet lamellar crystals with colouring from colourless and milky-white to blue and brown-yellow. Usually Celestine forms aggregates with radiant- or parallell located crystals or massive accumulations of dense or concretionary structure. Often forms pseudomorphosis on Calcite. Not too hard and heavy, Celestine possesses perfect cleavage, parallel bottom base of crystals. It is transparent more frequent, possesses glass and mother-of-pearl brilliance. Very dangerous.
Diagnostic indication.
Celestine it is sometimes possible to entangle with other sulfates, for example with barytes, whose specific gravity, however, considerably anymore. For verification it is possible to behavior a simple chemical test - to take powder of mineral on the tag of platinum needle and place in flame of gas burner. It will be painted in a carmine-red color due to the presence of strontium. A white, sky, or blue mineral consisting of strontium sulphate in orthorhombic crystalline form: a source of strontium compounds. Formula: SrSO4.
Origin provenance genesis.
Celestine can have a hydatogenesis. But for him sedimentary evaporite genesis is more typical. Jointly with him Sulphur is deposited, Sulfur, Brimstone, aragonite and complex of typical evaporite minerals.
Deposit minefield mine field occurrence subsoil.
Blue sky crystals are found in basalt ragstone Montekkio-madzhore (a province Vichenca) and in pegmatites of Madagascar. In areas Sicily and Romanya (Italy) is in rocks, containing gypsum and sulphur, Celestine usually white. Near Yena (Germany) it be found as veins of fibred structure of blue color. Deposit minefield mine field occurrence subsoil, suitable for industrial development, is in England, Russia and Tunis. Very large crystals, greyish-white and almost opaque, long to 75 sm and weighing to 2 kg found in the United States. Wonderful copies, though small sizes, are met in the Italian deposits. In Enne and Agrizhento (an area is Sicily, Italy) and in Pertikare (area to Brand) found out the magnificent groups (Druses) of crystals of celestine mostly practically white color (very light !!) in an association with by sulphur and gypsum.


Celestine is a sulfate of strontium in Druse (dangerous). Madagascar. A photo: © A.A. Evseev.
 Physical characteristics of celestine (celestite).
Physical characteristics of celestine (celestite).
This mineral can contain radio-active components, especially stone of intensive blue color (norm of natural radiation - 17-24 milliroentgen/hour). By the enhanceable level of radiation of stone and minerals is level of radiation from 29-32 milliroentgen/hour and higher. On the right is a sign of potential radiation danger (Ukraine, CIS, K.305). Transportation of large volumes of celestine without the sign of radiation danger is an image of "radioactive" (in Ukrainian, if CIS, in nuclear power plant and other) on the right - categorically forbidden is dangerous ore on strontium !!
Radio-active stone and minerals at the protracted carrying and uncontrolled application inward can cause the radiation damage of thyroid gland and cause its changes (including oncologic). Analogical damages can be caused the protracted carrying of dangerous minerals directly on a breast or in the pockets of masculine pantaloons. From the high background of radiation these dangerous for a health minerals are chosen by men, anchorwomen solitary way of life - and it is harmed this the health and surroundings (behavior the unhealthy way of life) - can "take" away disagreeable competitors celestine, including on a church (by the especially protracted reception of celestine inward in place of culinary salt - in the closed monasteries, sects and establishments of type of "magic").
Celestine (celestite) does not carry !!! In any case, from considering of safety and ecofriendlyness it is not recommended to carry poisonous and radioactive stone and minerals constantly and to hold the large standards of such minerals in an apartment or working cabinet (a house and apartment is a not mineralogical museum with the possible level of radiation from 32 to 120 milliroentgen/hour and higher for special exposition and mineralogical special safe, storage, depository, depot, storehouse, repository state institutions, where it is settled at presence of warnings denotations and special statements of workers of these specialized establishments). Celestny categorically forbidden expectant mothers and especially little children which uncontrolledly often pull interesting them objects in a mouth (in particular, interesting in appearance celestine and their accretions).
A radiation from a point source and small object decreases proportionally to the square of distance to this object. Stepping back on 2 meters from a dangerous object, you will decrease the level of study from this object in 4 times. Stepping back on 10 meters, you will decrease the level of radiation of this object in 100 times. If a point radiant has an object 2000 milliroentgen/hour at the natural background of radiation of environment 19 milliroentgen/hour (totally 2000 + 19 = 2019 milliroentgen/hour), stepping back from a dangerous object on 10 meters, you will secure itself to the level of radiation 20 milliroentgen/hour from an object and 19 milliroentgen/hour from an environment (total a general level of radiation from an object and environment will be 20 + 19 = 39 milliroentgen/hour).
Dangerous is a direct contact with a body and carrying on the body of point and diffuse poisonous and radio-active sources and components - especially water-soluble and hydatogenesis (an about 50% radiation is taken in at a contact with the external surface of body and an about 100% radiation - at a reception into a radio-active or infected object). Most dangerous is a direct reception into poisonous and radio-active components, stone and minerals, including in the pounded kind and especially soluble in a liquid. All of wilful injections poisonous and especially radio-active preparations categorically forbidden. It inedible and very dangerous for life and health stone and minerals !!
Use, practical application, deployment.
Celestine is basic ore on strontium which is used in pyrotechnics and production of tracings bullets (special project, not to breathe in smoke - strontium is painted by flame in a carmine-red color). Strontium is used also in nuclear energy, in the production of special varnish and other In home collections, keeping is not recommended.

Celestine. A kimberlite tube is Udachnaya, Yakutia. A photo: © A.A. Evseev.


Celestine. Sakoani, Madagascar. Zheoda of bluish crystals of celestine (Madagascar). A photo: © V.I. Dvoryadkin.


Celestine (blue, in emptinesses in a basalt). Montekkio-madzhiore, Vichenca, Italy. A photo: © A.A. Evseev.
Strontium is an element the exchange of which is related to the exchange of calcium. In an organism he is in amounts to 3-4 mgs in days and prevents development of caries and osteoporosis. Strontium, acting with food, relatively badly mastered an organism (about 5-10%). Absorption of strontium takes a place in a duodenum and iliac bowel (cancer of pancreas and spleen). The strontium absorbed in an organism hatches with urine (cancer of bud) and, in less degree, with a bile (cancer of hepar). There is the unabsorbed strontium in an excrement (cancer of colon).
In the organism of people mass 70 kg are an about 320 mg of strontium, thus his basic amount (to 99%) is deposited in bones. Relatively high concentrations of strontium - in lymphatic knots (0,30 ± 0,08 mcg/g), causes the cancer of lymphatic knots, in lights (0,20 ± 0,02), ovaries (0,14 ± 0,06), hepar and buds (0,1 ± 0,03). In blood found out 0,02 ± 0,002 mcg/ml of strontium. A lot of strontium are weak vital functions of organism (stimulus).
Strontium is able to accumulate in an organism. Middle maintenance of strontium in an organism is equal 0,002%. Basic displays of surplus of strontium: rickets-like diseases (curvatures of bones under own weight of people), "urovskaya" Kashin-Bek disease, endemic osteoarthritis, osteoarthritis deformans endemica; fibrosis of lights. For the leadingout of strontium from an organism use preparations of magnesium, calcium, food fibres, sulfate of natrium sodium and sulfate of barium. Celestine is cartilaginous pseudomorphosis on dead bodies (carcass).

Celestine on shells (pseudomorphosis). Mangyshlak, Kazakhstan. Middle Asia. A photo: © A.A. Evseev.
Substitution on 99,3% of cartilaginous making clamshell (Celestine into a shell, not pyrite)

Pseudomorphosis of celestine (sulfide of strontium). Mangyshlak, Kazakhstan. Middle Asia. A photo: © V. Ponomarenko.
Possible pseudomorphosis of celestine on cartilaginous fish and organic scale (alongside is aquatic caldera of volcano)
Calcium, being in composition bone tissues, on the properties near to strontium, therefore the ions of strontium can substitute for a calcium in bones. Thus there are cases of both synergism and antagonism of strontium. Vitamin of D, lactose, amino acid lysine and arginine improve absorption (recoveryс) of strontium. Vegetable food, rich in food fibres,, and also sulfate of natrium sodium and sulfate of barium, can diminish mastering of strontium.
"Urovskaya illness" (strontium rachitis) arises up because of expulsing of ions of calcium the ions of strontium from bone tissues or entering organism of strontium in place of calcium. An accumulation in the organism of strontium results in the defeat of organism, but typical is development of dystrophic changes of the osteoarticular system in the period of growth and development of organism, in youth (a symmetric deforming osteoporosis is formed from braking of growth of bones in the areas of metaphysial cartilages, there is not an area of growth of cartilaginous constituent of bone). Poisoning strontium.
Strontium provokes development of cartilage and connecting tissues actively (bones, copulas, joints, tissues of stomach). The consumption of ration with the lowered maintenance of strontium results in oppressing of growth, damages, surplus calciphylaxis of bones and teeth (enhanceable fragility, insufficient development of cartilaginous tissues and skin), increase of frequency of decay of teeth and fragility of nails (teeth and nails break a secret, fragile hairs, cut on ends). Eat is marrow (into beef and pork thigh-bone, femur) and pork legs (jellied, galantine), pork headcheese.
The radio-isotope of strontium of 90Sr is dangerous for an organism, which at a hit in the complement of bone tissues able to influence on marrow and violate blood-making hematopoietic processes (in place of provocation of growth of thigh-bone he destroys a radiation marrow inwardly bones, a sign is breaks of neck of thigh). Eat - bone flour, bones.
Food sources of strontium: bob plants (bobs, pea, soy, kidney bean and other), cereal (buckwheat, oat, millet, a wheat is soft, a wheat is hard, rice is wild, rye) and plants, formative root crops and tubers (ginger, potato, beet, carrot and other), fruit, are rich in strontium: apricots, quince, pineapples, vine, pears, kiwi; dried fruit: raisin, dried apricots, dates; nuts and seed: peanut, cashew nut, macadamia, a nut is Brazilian, pistachios, hazel filbert; greenery of greenery of celery, dill; algae: laminaria (oarweed); bones and cartilages.
DOPOG #7 

Radio-active materials (radiation, Ukraine)
Risk of absorption of external and internal radiation irradiation
To limit time of influence, burns by a radiation, radiation exposure of photo- and the cinema material
Yellow overhead half of rhombus, white - lower, isometric, number of DOPOG, black sign of radiation, inscription, title (text)
DOPOG #7 





Radio-active materials
Risk of absorption of external and internal radiation irradiation, radiation burn
To limit time of influence, burns by a radiation, radiation exposure of photo- and the cinema material
White, yellow overhead half of rhombus, white - lower, number of DOPOG, black sign of radiation, text
Active single-component (impoverished) radiation materials. White rhombus, one vertical red line down - there is not a nuclear military load (including simple h-bomb on a nuclear detonating fuse, impoverished uranium, burning war-head of "Kosovo", EU, single-component without a nuclear explosion). Jurisdiction is metrology of the Ukraine.
Yellow rhombus, two vertical red lines down is an active single nuclear charge (uranium, nuclear bomb on a type "Hiroshima", including multicomponent of the same type nuclear parts of single military load, single-component explosion and simple chain nuclear reaction - "two in one"). Jurisdiction is GAI of the Ukraine (Road Police Militia of the Ukraine) and war military law and order.
Yellow rhombus, three vertical red lines down - active double multi-component nuclear charge (plus heavy hydrogen-tritium, thermonuclear bomb of type the "Tihiy Pacific ocean", including nuclear parts of full-duplex military load, chain nuclear explosion and chain thermonuclear reaction - "three in one"). Jurisdiction - war military Road Police VAI of Ukraine and war military law and order.
DOPOG #6.1 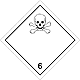
Toxic non biological matters substances (toxin)
Risk of poisoning at inhalation, contact with a derma or swallowing. Make a danger for a water environment or sewage system
To use a mask for emergency abandonment of transport vehicle
White rhombus, number of DOPOG, black skull and crossed cross-bones
DOPOG #5.1 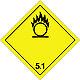
Matters substances, which oxidize
Risk of stormy reaction, self-ignition or explosion at a contact with combustible or flammable matters
To shut out formation of mixture of load with flammable or combustible matters (for example by sawdusts)
Yellow rhombus, number of DOPOG, black flame above by a circle
DOPOG #4.3 

Matters substances, which select flammable gases at a contact with water
Risk of fire and explosion at a contact with water.
Gruz, which had scattered, it is necessary to cover and hold dry
Sine-blue rhombus, number of DOPOG, black or white flame
DOPOG #9 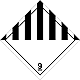
Other hazardous substances matters and produce
Risk of burns. Risk of fire. Risk of explosion.
Make a danger for a water environment or sewage system
Seven vertical black striaes on a white background is a top, white - lower half of rhombus, number of DOPOG
