Stone, minerals and semiprecious of the world stone
Sulphide: Molybdenite -->rus
 Diagnostic cart.
Diagnostic cart.
On a photo: standard from Arizona. Down: lamellar tabular Molybdenite on a dolomite
Mo S2
Crystal structure hexagonal
Hardness on the Mohs scale 1-1,5
Specific unit weight mass 4,6-5
Cleavage perfect absolute
Fracture, break uneven
Colors leaden black
Colors in powder triturate greenish-grey
Glance (glitter, glare) metallic
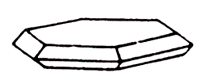
Molybdenite (molybdenum brilliance) is a sulfide of molybdenum. Glance (glitter, glare) metallic, opaque. Colors leaden-grey. Line also leaden-grey. The thin-растертый has a greenish tint. Flexible, but not elastic. Cleavage very perfect absolute. Genetically related to the granites, greisenи, quartz vein, lode, mines, rarer with pegmatites. Crystals (hexagonal Crystal structure) of plate tablet lamellar. More frequent scaly, fissile or plate tablet lamellar aggregates. Main base ore of molybdenum. Deposit minefield mine field occurrence subsoil: in the USA, Canada, Chile, Norway, CIS.
Molybdenite is a soft grey mineral consisting of molybdenum sulphide in hexagonal crystalline form with rhenium as an impurity: the main source of molybdenum and rhenium. Formula: MoS2.
Molybdenite is presented the scaly tablets of hexagonal outlines. Scales are often bent or twisted up and does not have the clear expressed crystalline outlines. A mineral is opaque, a color is leaden-grey bluish, with metallic off-gloss; it is heavy, often fat by touch, possesses perfect cleavage on plate tablet lamellar. In thin plates flexible, but not elastic.
Chemical composition (chemistry, compound). Molybdenum (Mo-permalloy) 60%, Sulphur, Sulfur, Brimstone (S) 40%. Form of crystalline excretions. Well well-educated crystals are presented hexagonal tablets; in most cases Molybdenite is presented fissile and scaly aggregates and spherulites. Crystalline structure. Typical stratified lattice. The flat nets of crystalline lattice, formed the layers of molybdenum, are disposed between two layers of ions grey, parallell to the basale plane. Class of symmetry. DiHexagonal-bipyramidal. Cleavage. Perfect on a base (0001). Aggregates. Dense, thin-lamellar.


Diagnostic indication.
On a hexagonal form and very low hardness of Molybdenite it is possible to entangle with a graphite. A grey, pounded line acquires a spring onions-green color (the line of graphite remains grey). Distinguishing them is possible on a line on a paper or on the porous unglazed porcelain; a graphite gives a black-grey gray line, and Molybdenite - greenish-grey gray. Not melt fuse. A behavior is in acids. Hardly decomposes in the concentrated sulphuric acid at the slow boiling.
Origin provenance genesis.
Enough ordinary and widespread mineral; be found mainly in high temperature hydrothermal vein, lode, mines. Associated with the minerals of tin, copper and tungsten. Often be found in granites and pegmatites.
Deposit minefield mine field occurrence subsoil.
In Italy Molybdenite obtain in many places. Good standards take a place from Arendalya in Norway, Azegura in Morocco, state Queensland in Australia and, mainly, from the districts of Temiskaming and Oldfil'd-Taunship in a province Quebec (Canada). The deposit of Klaymaks in the state of Colorado (USA) is developed by an industrial method.
Use, practical application, deployment.
Из Molybdeniteа извлекается молибден, используемый в основном для получения особых сплавов. Благодаря своей очень малой твердости и способности распадаться на мельчайшие жирные частицы Molybdenite находит применение как сухая смазка, особенно при высоких температурах.



From Molybdeniteа a molybdenum, in-use mainly for the receipt preparation of the special alloys, is extracted. Due to the very small hardness and ability to disintegrate on the shallow fat particles of Molybdenite finds application use as a dry greasing, especially at high temperatures.
A molybdenum operates as a cofactor in composition enzymes, accountable for detoxication of organism, has influence on the use supplies of iron in an organism, activates the exchange of sulfur-containing amino acid for work of the nervous system and brain (psychiatry), instrumental in withholding of fluorine in an organism, strengthening the enamel of teeth.
Molybdenum from food products and in form soluble complexes absorbed. For people 25-80% is sucked in molybdenum, acting with food. Absorption takes a place in a stomach and on all of thin bowel, in a greater degree in its proximal department, what in distal ("complex of dead body"). On suction of molybdenum co-operations influence between a molybdenum and different dietary (food) forms grey (Molybdenite).
An about 80% molybdenum, entering blood, contacts with squirrel (with an albumen) and transported on an organism. In blood a molybdenum is distributed evenly between uniform elements and plasma. Greater part of molybdenum gets in buds and hatches by them. An egestion is the main mechanism of his homoeostatic adjusting. Substantial amounts are excreted with a bile. The accumulation of molybdenum does not take a place in an organism (except for the brain of people).
A molybdenum is needed: at new formations of cerebrum (tumours), paropsiss, tachycardia, masculine fruitlessness. Consider that a tungsten, lead and natrium sodium, operate as antagonists of molybdenum and cause his deficit in an organism. Signs of insufficiency of molybdenum: hypererethism, crabbiness, dysaptation ("dark"), "chicken blindness", violation of rhythm of heart-throbs (tachycardia), increase of risk of development of shrine of gullet, decline of activity of enzymes, containing a molybdenum.
The deficit of molybdenum is possible for people, recipient a complete parenterally feed or subject to stress (a megascopic requirement is in sulphiteoxidase). For patients, recipient the protracted parenterally feed, the syndrome of the "purchased deficit of molybdenum is described": hypermethioninemia, hypoiricosuria, hyperoxipurinemia, hypo uricosuric and hyposulphaturia, making progress mental (cerebral and cerebral nervous) disorders (to the coma are the urgent states). Organs, containing the high amounts of molybdenum - hepar and buds.
Basic displays of surplus of molybdenum: an increase of activity of xanthine oxidase, increase of level of urinary acid, is in urine; gout (uraturia, urolithiasis); irritation of mucous membranes, black-lung disease pneumoconiosis; oppressing of blood formation, hematopoiesis, hematogenesis (anaemia, leukopenia), decline of mass of body. At a surplus consumption - within the limits of 10-15 mg/day - the clinical symptoms of intoxication show up. At the doses of molybdenum, exceedings 15 mg/day , activity of xanthine oxidase rises, urinary acid accumulates, the risk of gout is increased (at persons, contacting with a molybdenum in a production). Symptoms, which show up the irritation of mucous membranes, black-lung disease pneumoconiosis, degrowth body, develop during chronic molybdenum intoxication.
DOPOG #8 
Corrosive (caustic, pungent, acrid) substances matters
Risk of burns as a result of eating away of derma. Can stormily react between itself (components), with water and other matters. Matter, that spilled / scattered, can select a corrosive pair.
Make a danger for a water environment or sewage system
White overhead half of rhombus, black - lower, isometric, number of DOPOG, test tubes, hands
